Not all progesterone’s are created equal
A review of progesterone and breast cancer risk by Dr. Nyjon K Eccles, BSc MBBS MRCP PhD
Dr. Nyjon Eccles is known as ‘the natural doctor’ and practices from Harley Street in London, England and is one of the UK’s leading Integrated Medicine doctors with a special interest in breast cancer.
His article is a summary of some of the evidence surrounding the use of natural progesterone vs. synthetic forms and it seeks to clarify the debate as to whether all types of progesterone have the same physiological and clinical effects, or whether there is indeed evidence to suggest that the synthetic progesterone’s (progestins) differ in effect to progesterone in its native molecular form.
Note: From herein progestins will refer to the synthetic form and progesterone to the natural form.
I am indebted to the commentary and detailed review published by Dr. Kent Holtorf in 2009. Much of his article is so pertinent to this discussion that I have pasted in some sections of it. Some of this is detailed science for those who have the appetite for it, but I have also highlighted the key conclusion points for patients in the summary at the end.
A lot of confusion abounds on this topic, and in my experience most doctors are confused about this. As with all truth seeking, this can only come from a critical look at the published data and not from mainstream media or expressed opinions that do not reference the published science. The addition of synthetic progestins to estrogen in HRT for menopausal symptoms, increases the risk of breast cancer more than estrogen alone (1).
In breast cancer survivors, progestin use is associated with an increased breast cancer risk compared with its non-use (2). However, outside pregnancy, natural progesterone whether endogenously produced or exogenously administered does not have a cancer-promoting effect on breast tissue. In postmenopausal women, progesterone is added to prevent the carcinogenic effect of estrogen on the uterus (3). In premenopausal women, the potency of the progestin in most oral contraceptive pills appears adequate to provide a protective effect against endometrial cancer.
Progestagens counteract the adverse effect of estrogens on the endometrium (see figure 1), the effect being greater the more days every month that they are added to estrogen and the more obese that women are (3).
Figure 1: The position of the endometrium in a woman’s uterus.
The notion that progesterone may increase breast cancer risk is based on a secondary follow-up of a French study (4) investigating breast cancer incidence in approximately 80,000 women who had estrogen and various progestogens for hormone replacement therapy. It was suggested that progesterone increases breast cancer risk, contrary to popular thinking that this hormone is safe and protective against breast cancer.
Fournier et al’s (5) original study looked at the relationship of different progestogens (any molecule with a structure like the natural hormone progesterone that binds to and activates intracellular progesterone receptors), in combination with estrogens, on the risk of developing breast cancer. In their first study they found that of all the progestogens studied, natural progesterone had the lowest risk, and that this risk was a lower risk than no treatment at all. (2)
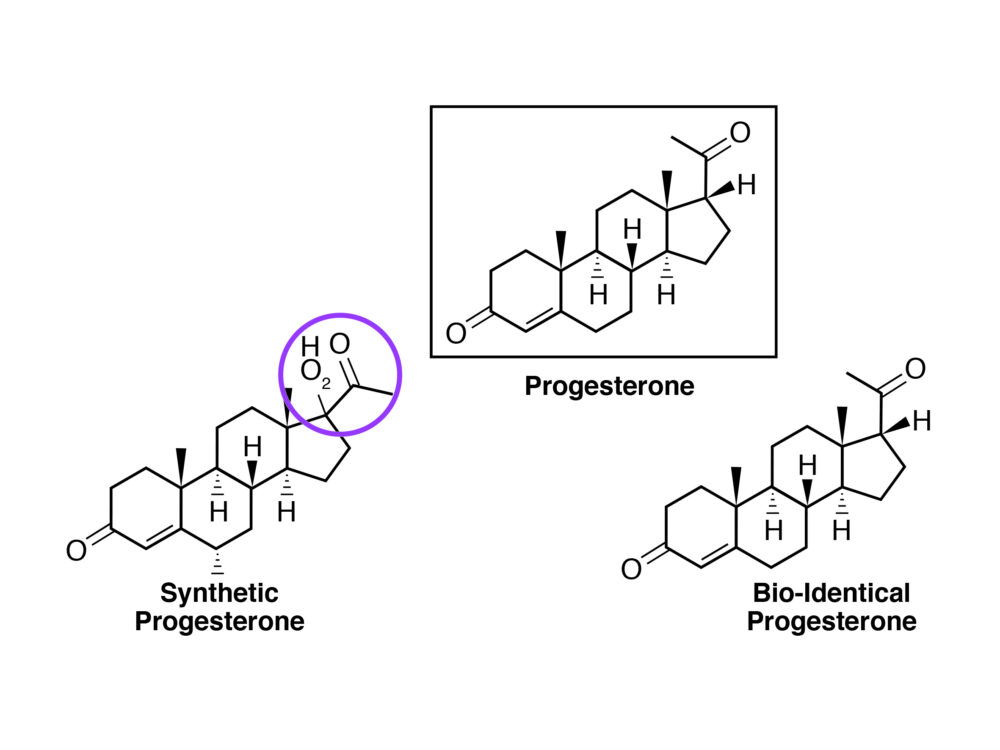
Figure 2: The difference between a synthetic progesterone (progestin) and a natural (or bioidentical) progesterone.
Their second study (5) in the same patient population as the first study (80,391 postmenopausal women), showed that the risk with estrogen plus progesterone is less than the risk of estrogen alone (1.7 vs 2.1, respectively), and that of all the progestogens, natural progesterone has the lowest risk.
This study did not look at the effect of natural progesterone by itself–only estrogen plus progesterone. A more accurate commentary on their data is that natural progesterone decreased the risk of breast cancer caused by long-term use of estrogens (i.e., risk 2.1 to 1.7).
These studies were based on the use of oral progesterone. Women who have an excess of estrogen relative to progesterone (low progesterone/estradiol ratio), are more likely to have atypical benign breast disease which carries increased risk of developing into breast cancer. (6)
Low endogenous luteal progesterone levels in premenopausal women (much more prevalent in peri-menopausal woman), have also been associated with increased breast cancer risk (7). One small study (8) looked at the risk of breast cancer with topical progesterone (10-30 mg progesterone daily). This showed the breast cancer risk to be reduced by half in those using topical progesterone for 3 years or more.
Estrogen and progesterone receptor positive cancers
About 70% of breast cancers are ER+ (estrogen receptor-positive), and most of these breast cancers (about 87%) are also PR+ (progesterone receptor-positive). Hormone receptor status is a significant factor in considering breast cancer treatment. There is a general view that having a breast cancer that is both ER+ and PR+ may be worse than having ER+ alone.
Paradoxically, women with high levels of both estrogen and progesterone receptors (high ER+ and PR+ status) often have the best chance of surviving. Alas, this information is often not passed on to patients. Whereas estrogen can promote a tumor’s growth, progesterone slows growth. Estrogen and progesterone receptors are proteins found in many of our cells, including cells in the breasts. Both receptors are directly involved in switching some 470 genes on and off, thereby affecting cell behaviour. While estrogen activates its receptor, turning on genes that stimulate cells to keep dividing, driving tumor growth, sufficient progesterone on the other hand will slow down the estrogen-fuelled growth and division of these cells.
The late Dr. John Lee, MD, author of What Your Doctor May Not Tell You About Breast Cancer, detailed this years ago. He maintained that when activated by progesterone, the progesterone receptors attach themselves to the estrogen receptors, stopping estrogen from turning on genes that promote the growth of the cancer cells. Progesterone activates genes that promote death of cancer cells (apoptosis) and the growth of healthy, normal cells.
A failure to grasp this important concept has led to many doctors ‘villainizing’ progesterone and progesterone status. A study published in the highly respected scientific journal Nature, led by Cambridge-based Cancer Research U.K. researcher Dr. Jason Carroll of the University of Adelaide in Australia, brought more awareness to the benefits of progesterone and progesterone receptor-status, (3) [Phil, this sentence is very confusing. Is Dr. Carroll from Cambridge or the U of Adelaide?—and the reference # does not list Carroll as one of the authors, nor is it from the cited journal (Nature)]
and is a reminder that the presence of both ER and PR status has typically been considered an indication of how good a woman’s chances of surviving were. The belief being that these cancers were more ‘treatable’ than hormone receptor-negative cancers. Carroll’s study found that progesterone – via the progesterone receptor – moderates how the estrogen receptor works. They found that the progesterone receptor, in effect, ‘re-programs’ the estrogen receptor, changing the genes that it influences. (For further details see Nature, volume 523, pages 313–317, 16 July 2015). This study highlights an important function for the PR receptor in modulating the behaviour of the ER in breast cancer. It confirms the previously published work that has suggested the same effect.
It is most important to note that the overall effect of progesterone on cancer cells was to cause the cells to stop growing as quickly. Carroll’s findings clarify why women who have both ER+ and PR+ potentially have a better outlook than those with just ER+ or receptor-negative cancers; assuming, that is, that progesterone forms part of their treatment regime. Progesterone and HER2 in Breast Cancer HER2 and progesterone seem to be important in controlling metastatic dissemination of tumor cells prior to the detection of a primary tumor.
Researchers have known for some time that synthetic progestins, unlike natural progesterone, do not stimulate activation of the tumor suppressor gene p53 when it attaches to progesterone receptors. (4) P53 is a repair gene, which protects cells from cancerous change if progesterone can attach itself to progesterone receptors.
Maintaining healthy natural progesterone levels, avoiding synthetic progesterones and the downregulation of HER2 seems to be a desirable treatment objective, and whilst Herceptin® is the drug of choice for HER2, one author has noted that daily consumption of 25 grams of flaxseed has been shown to decrease HER2 expression by 71%, which appears to outperform the drug, without the side effects of the drug.
Summary
- The use of synthetic progestins in breast cancer survivors is associated with increased risk of recurrence, whereas natural progesterone use does not increase risk.
- The addition of progesterone to estrogen negates the increased risk of breast cancer seen with estrogen alone.
- Women who have an excess of estrogen relative to progesterone (low progesterone/estradiol ratio), are more likely to have atypical benign breast disease and increased risk of developing breast cancer.
- Women with breast cancers with high levels of estrogen receptors and progesterone receptors have the best chance of survival.
- Sufficient progesterone will slow down the estrogen-fuelled growth and division of breast cancer cells.
- Natural progesterone, (but not synthetic progestins), activate genes that promote death of cancer cells (apoptosis) and the growth of healthy, normal cells.
- Carroll’s study published in Nature in 2015 showed that progesterone acts as a suppressor of estrogen stimulated breast cancer cells.
- P53 is a repair gene, which protects cells from cancerous change if natural progesterone can attach itself to progesterone receptors. This effect is not seen with progestins.
Yet, despite all these positive facts, the addition of natural progesterone to estrogen receptor modulation is not currently standard oncological practice.
References
- Jerry DJ. Roles for estrogen and progesterone in breast cancer prevention. Breast Cancer Res. 2007, 9: 102-10.1186/bcr1659.
- Beral V, Million Women Study Collaborators. Breast cancer and hormone-replacement therapy in the Million Women Study. Lancet. 2003, 362: 419-427. 10.1016/S0140- 6736(03)14596-5.
- Beral V, Bull D, Reeves G, Million Women Study Collaborators. Endometrial cancer and hormone-replacement therapy in the Million Women Study. Lancet. 2005, 365: 1543-1551. 10.1016/S0140-6736(05)66455-0.
- Fournier et al J Clin Oncol 26 (8):1260-1268, 2008. [Needs author’s initial, and article title]
- Breast Cancer Res Treat 107: 103-111, 2008. [Author, title?]
- Sitruk-Ware et al. J Clin Endocrinol Metab 44, 771, 1977. [Needs article title]
- Micheli et al. Int J Cancer 112, 312-318, 2004. [Needs author’s initial, and article title]
- Plu-Bureau G, et al. Cancer Detect Preve 23(4), 290-296, 1999. [Needs article title]
