Vitamin B17: An Alternative Cancer Treatment
If you’re a baby boomer or older, you may remember hearing about Laetrile, the highly publicized cancer drug that provoked an unprecedented medical and political controversy that peaked in the 1970’s. Proponents considered Laetrile, a synthetic form of the naturally occurring plant compound amygdalin (derived from apricot kernels), a safe and “natural” alternative to conventional toxic chemotherapy, but the medical establishment called it a “fraud,” dismissing it as an ineffective and toxic drug that should be prohibited from use. A series of mostly negative animal studies from the 1970’s and a clinical trial in the early 1980’s that came up empty put the lid − perhaps too soon − on further human testing of this substance which never gained FDA approval. But interest in amygdalin has been revived in recent years, with new studies on cell cultures revealing previously unknown mechanisms of action against a variety of cancers.
Up until now, those wishing to take advantage of amygdalin’s purported benefits could only do so by consuming apricot kernels or similar pills of unknown purity and potency. But now, IAS offers a superior alternative: B17-Pro™ which is purified amygdalin in 100 mg tablets. In this article, we’ll cover the chemistry and properties of amygdalin, its eventful history, the contentious relationship between the medical establishment and amygdalin (Laetrile) proponents, the pros and cons of its use, new discoveries and potential applications, and its use today.
Intro: An Amygdalin (Laetrile) Primer
Amygdalin, derived from the Greek word meaning “almond,” is a naturally occurring substance found in the pits, seeds, and kernels of hundreds of fruits and plants such as apricots (the richest source), bitter almonds, apples, peaches, plums and berries. Amygdalin belongs to a class of compounds known as cyanogenic glycosides, (containing one or more simple sugar molecules and a nitrile (−C≡N) group that can potentially be metabolized in the body to hydrogen cyanide (HC≡N)). Natural amygdalin is an optically active isomer with the chemical name D-mandelonitrile-beta-D-gentiobioside, also referred to as D-amygdalin, (see figure 1). US-patented Laetrile (a synthetic derivative of amygdalin known as L-mandelonitrile-beta-D-glucuronide), is structurally and pharmacologically like amygdalin (see figure 2) and technically also a cyanogenic glycoside. Both compounds are also called nitrilosides.
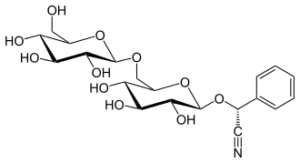
Figure 1: Amygdalin (D-mandelonitrile-beta-D-gentiobioside)
Figure 2: US-Patented Laetrile (L-mandelonitrile-beta-D-glucuronide)
Amygdalin and its synthetic derivative have been promoted as alternative cancer treatments since the early 1950s.
It’s impossible to discuss one without the other, since they are related substances whose names are used interchangeably in the medical literature and elsewhere, but technically they are not identical. But at some point, after it gained patent in the US in 1961, the term “Laetrile” became synonymous with “amygdalin.” This merging of terms, as one author put it; “confuses an already complicated story.” (1) A research paper from the 1970’s went on to say that; “amygdalin at various concentrations and with numerous impurities is the most common cyanogenic glycoside found in Laetrile samples.” (2) Considering that US-patented Laetrile was probably never manufactured on a large scale, and that Mexican-produced Laetrile is D-mandelonitrile-beta-D-gentiobioside, i.e. amygdalin, made from crushed apricot pits, (3) it seems safe to assume that the chemical identity of the substance used by “Laetrile” practitioners and studied in animal and human trials was and currently is amygdalin. To add to the nomenclature confusion, “Vitamin B17” has also been applied to both Laetrile and amygdalin (we’ll see more on B17 later).
[Note: In this article, as much as possible, especially when discussing the scientific studies, we’ll refer to the substance D-mandelonitrile-beta-D-gentiobioside as “amygdalin.” When discussing its historical significance, as in the next section, and its use by medical practioners, we’ll use the term “Laetrile,” since this was the designation commonly applied to this therapy starting in the 1960’s.]
The main proposed anti-cancer mechanism is localized delivery of cyanide to cancer cells, destroying them. When amygdalin interacts with the enzyme beta-glucosidase and water, it degrades to produce hydrogen cyanide, benzaldehyde, and glucose. (2,4,5) Cyanide release was originally thought to be the main cancer-fighting mechanism, (4,6) but benzaldehyde and prunasin (another breakdown product of amygdalin) may also inhibit cancer cells. (4,6,7) In fact, benzaldehyde has been shown to be effective as a primary cancer therapy in humans. (7)
The usual course of Laetrile treatment involves intravenous (IV) administration by a physician for a period (several weeks or months, according to response) followed by oral maintenance therapy (tablets), usually as part of a comprehensive “metabolic” program consisting of a specialized plant-based diet, high-dose vitamins and pancreatic enzymes.
The main risk of Laetrile therapy (and a major aspect of its controversial use) is cyanide toxicity, especially when administered orally. Intestinal bacteria and some commonly consumed plants contain enzymes, (beta-glucosidases) that initiate the breakdown of amygdalin and cleave the nitrile group, releasing cyanide into the bloodstream. (4) Oral amygdalin is estimated to be 40 times more potent than the IV form due to its enzymatic conversion to cyanide in the GI tract. (8) In contrast, IV administration bypasses the GI tract, limiting rates of conversion to cyanide. (9)
Proponents consider amygdalin (Laetrile) an effective and natural alternative to conventional side-effect-ridden chemotherapy, with decades of safe use if administered by an experienced practitioner, while the conventional medical establishment has, over the past five decades, predominantly labeled it a “fraud,” a “cancer quackery,” ineffective, and potentially toxic.
Laetrile has been banned in the US and the EU for decades, but amygdalin-rich apricot kernels and apricot-based pills are available. However, amygdalin tablets (B17-Pro™), consisting of the isolated active ingredient, must be purchased online. Amygdalin and Laetrile are still permitted to be manufactured and administered in clinics in certain countries, such as Mexico.
History, Controversy, and Politics
Amygdalin, in the form of bitter almonds, has been used for centuries. It was first isolated in France in 1830, and was later used to treat cancer in Russia in 1845 and in the US in the 1920’s. In the 1950’s, an IV form was synthesized and patented as Laetrile in 1961. In the years following, the public’s quest for a “natural” alternative to harsh chemotherapies caused Laetrile’s popularity to soar − it’s estimated that by 1978, more than 70,000 Americans had undergone treatment with it. (9,10)
But the 1970’s was a decade characterized by an unprecedented medical, political, and even ethical controversy as Laetrile’s availability to the public became threatened when an Investigational New Drug (IND) application was rejected by the FDA based on lack of efficacy in animal studies. (In the US, an IND application must be filed with the FDA prior to clinical studies in humans as part of the drug approval process.) Even though the FDA is charged with ensuring that any drug on the market is both safe and effective, the agency’s thumbs down on Laetrile was seen by many as a violation of one’s “freedom of choice” in determining his or her choice of cancer treatment. Laetrile supporters believed that the FDA, the pharmaceutical industry, and the medical establishment (including the American Medical Association and the American Cancer Society) were intentionally conspiring to restrict access to a potentially life-saving drug. One of the top centers of cancer research in the US, the Memorial Sloan-Kettering Cancer Center in New York, was even suspected of covering up positive results in animal studies (we’ll see more on this later).
Legal proceedings in Oklahoma, Massachusetts, New Jersey, and California challenged the FDA’s authority, which led to the legalization of Laetrile in 27 states in the 1970’s. But in 1980, the US Supreme Court upheld a Federal ban on interstate shipment of Laetrile, which effectively put an end to its use in the US. Laetrile never gained approval by either the FDA or the European Commission. However, as mentioned, it is still manufactured and administered in clinics, predominantly in Mexico, but also elsewhere around the globe.
Rationale for Amygdalin (Laetrile) Use
Supporters of amygdalin (Laetrile) base their claim on its anti-cancer properties largely on epidemiological data, citing indigenous peoples such as the Hunzakut tribe of the Karakoram Mountains of northern Pakistan who routinely consume nitrilosides and typically live to 100 years old with very low rates of cancer. (The assumption here is that nitriloside intake, including amygdalin, is directly responsible for longevity and low-cancer incidence, but other factors such as a healthy diet, ample exercise, and a rich social network also contribute to these peoples’ health.) And as far as epidemiological observations, though they are informative, they are not proof of efficacy.
One of the main early promoters of amygdalin (Laetrile), biochemist E.T. Krebs Jr., labeled it “vitamin B17,” and believed it to be a natural cancer-fighting food component, that if regularly consumed, as in the Hunzakut diet, would prevent various types of cancer. According to Krebs, cancer is a vitamin B17 deficiency disease. However, “vitamin B17” does not meet the definition of “vitamin,” is not required for normal metabolism, and is not an approved designation.
Besides epidemiological data, Laetrile proponents also point to case reports from doctors who have apparently administered it safely, effectively, and without toxicity for decades. But case reports are difficult to interpret due to lack of uniform documentation, use of conventional chemotherapies in combination with Laetrile, and variations in the dose and duration of treatment. (3)
As mentioned, cyanide is thought to be the main anti-cancer agent formed from amygdalin metabolism, and the rationale among Laetrile practitioners is to maintain cyanide blood levels high enough, but still below the toxic threshold to ward off cancer and boost the immune system. According to one doctor with a long history of using Laetrile in his practice, Dr. Philip E. Binzel, Jr., “Laetrile is nothing more than a concentrated form of nitrilosides. When ingested, nitriloside has merited recognition by maintaining non-toxic cyanide levels and acting as a potential threat to the immune surveillance, thereby lessening the frequency of cancerous tumors.” (11)
Various mechanisms have been proposed to explain amygdalin’s purported anti-cancer activity. One is that it is preferentially toxic to malignant cells, since they exhibit higher activity of beta-glucosidase than normal cells and lower levels of another enzyme, rhodanese (thiosulfate sulfurtransferase). (2,3) Beta-glucosidase initiates the breakdown of amygdalin, producing cyanide, and rhodanese converts cyanide to the non-toxic substance thiocyanate. (3) A higher ratio of beta-glucosidase to rhodanese in cancer cells favors increased cyanide production with lower rates of conversion to thiocyanate, making the drug much more lethal to cancer cells than normal cells. Another proposed mechanism involves another altered enzyme ratio, this time between beta-glucuronidase (not to be confused with beta-glucosidase) and rhodanese, which also leads to higher localized cyanide concentrations in cancerous cells. (3) Some studies do demonstrate the difference in concentrations of the enzymes rhodanese, (12) beta-glucosidase, (13) and beta-glucuronidase (14) in normal versus malignant tissues. However, other studies have found that cyanide is also released in normal cells, (15) and highly elevated oral doses have led to cases of deaths and toxicity. (2)
One cancer-inhibiting mechanism less often proposed by Laetrile proponents − but positively demonstrated in studies − is the amygdalin breakdown product benzaldehyde, which has shown efficacy in humans against tumors. (7)
Does it work? What the Studies Show
As mentioned, the conventional medical establishment has flatly rejected amygdalin (Laetrile) as a potential chemotherapeutic agent and most studies from the 1970’s and early 1980’s found it ineffective against a variety of cancers, in animals and humans, and potentially dangerous due to cyanide toxicity. Upon reviewing the medical literature, it’s common to find conclusions such as this one, from a panel of independent reviewers: “Laetrile has shown little anti-cancer activity in animal studies and no anti-cancer activity in human clinical trials.” (3) And this one, from a Cochrane review: “The risk-benefit balance of laetrile or amygdalin as a treatment for cancer is therefore unambiguously negative.” (8)
But it should be mentioned that the studies − animal and human − are laced with inconsistencies and ambiguities, and often present conflicting data, of which we’ll see numerous examples below.
Animal studies
Animal studies from the 1970’s evaluating the potential cancer-inhibiting activity of amygdalin reported predominantly negative findings, including two from 1975 sponsored by the National Cancer Institute (NCI). In these studies, various cancers were transplanted into mice and rats (osteogenic sarcoma, melanoma, carcinosarcoma, lung carcinoma, and leukemia). The rodents were then administered either amygdalin alone or in combination with beta-glucosidase (presumably to enhance the degradation of amygdalin to cyanide). Neither study found any significant cancer-inhibiting activity in any of the tumor types or leukemias studied with either the single or combination treatment, and both showed that co-administration with beta-glucosidase led to increased toxicity, (16,17) an expected result due to the higher rate of conversion to cyanide. Although other animal studies from the 1970’s using transplanted tumors corroborated these negative findings, (18,19) one study did report success in treating breast cancer in mice with combination therapy (amygdalin, vitamin A and enzymes). (20)
Other positive results were the originally hushed-up and later publicized set of unpublished animal experiments performed at Memorial Sloan-Kettering Cancer Center in the 1970’s in which amygdalin was shown to reduce the spread of cancer. These studies were performed by respected researcher Dr. Kanematsu Sugiura on mice bred to spontaneously develop tumors (not transplanted tumors as in the NCI studies). Sugiura found that amygdalin was 80% effective in preventing the spread of lung cancer in treated mice compared to a control group. (21) Apparently, Sloan-Kettering covered-up these results (for various reasons, perhaps political or economic) until they were leaked to the press by a principled and forthright employee − the subject of a documentary film; “Second Opinion: Laetrile at Sloan Kettering.” (21)
Human Studies
Amygdalin (Laetrile) is an unusual case; it never gained IND approval from the FDA (based on negative findings in animal studies), a preliminary step to proceed with clinical trials, yet the NCI agreed to sponsor (and the FDA approved) phase I and II clinical trials nonetheless, bypassing this regulation and most likely responding to public pressure. Prior to the clinical trials, the NCI attempted to compare the efficacy of Laetrile versus conventional chemotherapy by reviewing case reports of patients treated with each method. The problem was, the review panel couldn’t obtain sufficient data on Laetrile − after sending out close to half a million requests to doctors for information on their patients who had benefitted from the drug, the NCI received a meager 93 responses, and many case reports had to be thrown out due to insufficient documentation. In the end, the panel of 12 oncologists, blinded to the actual treatments, evaluated 160 cases (68 Laetrile, 68 chemotherapy, 24 controls). Of the 68 Laetrile cases, the panel found only two complete and four partial responses to the drug, precluding any definite determination as to its efficacy. (10) Yet, the NCI agreed to sponsor clinical trials.
The 1981 phase I trial was a very small study on only six patients that evaluated the safety of oral and IV administration in doses like those commonly used by Laetrile practitioners. Neither the IV nor the oral dose (500 mg tablet, three times daily) produced any toxic reaction, though the tablets did raise blood cyanide levels and led to symptoms of toxicity in one patient who also consumed a large helping of raw almonds. The researchers concluded that; “amygdalin in the doses employed produces few clinical side effects,” i.e., minimal toxicity. (22)
Since amygdalin was shown to be reasonably safe in the phase I trial, a larger phase II efficacy trial was conducted at the Mayo Clinic in 1982. 178 patients with various types of cancer received amygdalin intravenously for 21 days, followed by oral tablets (500 mg, three times daily), using doses and schedules “representative of current Laetrile practice.” The IV vials used in the study contained DL-amygdalin, a racemic mixture, which is a combination of both natural D-amygdalin (the biologically active isomer) and L-amygdalin (an artificial, inactive form) “so that the preparations would correspond with the products distributed by the major supplier.” The oral tablets contained only D-amygdalin. In keeping with the comprehensive metabolic therapy program used by Laetrile practitioners, patients also received vitamins and pancreatic enzymes, and followed a specialized diet. Of 178 patients, only one responded to treatment. And even though amygdalin produced minimal side effects in the previous safety study, several patients in this trial experienced symptoms of cyanide toxicity (granted, this was a much larger study). The researchers concluded that “amygdalin (Laetrile) is a toxic drug that is not effective as a cancer treatment and does not warrant further study” (23). It conformed to the overriding view of the medical establishment at the time and ended the prospect of any further human testing some 35 years ago.
Problems with the Phase II Study
This last study has been criticized by Laetrile supporters and there are several points of contention, one of which is the type of amygdalin used for IV administration. As mentioned, D-amygdalin is the biologically active isomer, and this was the substance used in the oral tablets. But the IV vials contained a racemic mixture, a combination of the D and L isomers (one active, one inactive), which would have significantly lowered the potency (by approximately 50 percent). The authors, as stated above, attempted to simulate the product characteristic of the Mexican supplier at the time, but it’s impossible to know decades later if this was an accurate simulation. It makes sense that the active isomer, D-amygdalin, was used in the oral tablets, but why was the racemic mixture used in the IV vials?
Another criticism is the “one size fits all” schedule of IV and oral doses used in the study. All patients were automatically switched from IV therapy to oral tablets after three weeks, regardless of response. Although the study authors stated that “the dosage and schedule were representative of past and present Laetrile practice,” (23) many practitioners generally advocate an individualized protocol, with the IV portion performed until a positive response is seen, often from four to 10 months, or even longer. (24) Patients are then put on oral maintenance (tablets) only when their cancer is in check and put back on IV if the cancer re-appears or grows. Some practioners even administer oral tablets while tapering off IV.
Perhaps the correct conclusion to be drawn from this study isn’t that “amygdalin (Laetrile) is ineffective as a cancer treatment,” (23) but rather, that administering a drug of questionable potency on a truncated schedule to advanced cancer patients doesn’t work.
Recent Studies
Although the conclusion of this last 1982 study precluded any further human trials, leaving the question of amygdalin’s efficacy in humans unanswered, the passage of a few decades − and some distance from the various political and scientific disputes − has led to renewed interest in recent years, and studies using cell cultures have resulted in promising findings.
Amygdalin has been shown to induce apoptosis (programmed cell death), (15,25) among other important mechanisms, and inhibit various types of cancer in breast, lung, bladder, colon, prostate, liver, kidney, and cervical cell lines. (15, 25-32) All of these studies draw a favorable assessment of amygdalin’s cancer-inhibiting potential. For example, a 2018 study concludes: “This compound might be applicable in the treatment of various cancer cell types,” (15) quite the opposite determination from the majority of studies from the 1970’s and early 1980’s. So, the door has not been closed on amygdalin after all, as new research is uncovering potential applications and previously unknown mechanisms of action.
Amygdalin’s Use Today and How to Use B17-Pro™
As mentioned earlier, amygdalin (Laetrile) can be legally administered in clinics only in certain countries. Dr. Antonio Jimenez, chief medical officer and founder of Hope4Cancer Institute in Mexico is one of the doctors who uses it in his practice. Besides its role as a chemotherapeutic agent, Dr. Jimenez has observed that amygdalin provides pain relief and well-being enhancement in his patients. He doesn’t use it as a single therapy or “magic bullet,” though, but as part of a comprehensive treatment program to slow the progression of cancer (Dr. Jimenez has a realistic expectation, acknowledging amygdalin’s potential to inhibit cancer, but also realizing that it alone is not a cure, but an integral part of a multi-pronged approach to treatment.)
Outside of Mexico, those in the US and most European countries don’t have access to clinics such as the Hope4Cancer Institute, but instead resort to consuming apricot kernels or apricot-based pills (with unknown amounts of amygdalin of unknown purity). A superior alternative is B17-Pro™, containing 100 mg amygdalin per tablet, plus pancreatic enzymes and zinc. The recommended dose is one tablet per day with water before a meal, or as directed. Consult your health care professional before using. Remember not to take amygdalin tablets shortly before eating raw almonds or crushed fruit pits, or with fruits or vegetables that contain beta-glucosidase or beta-glucuronidase (i.e., celery, peaches, bean sprouts, carrots, etc.)
Dr. Jimenez apparently has never seen a toxic reaction with the IV and oral doses he uses in his clinic, and that observation is shared by other Laetrile practitioners. But, as we saw earlier in the studies, cyanide toxicity is a concern. Symptoms include liver damage, difficulty walking, fever, coma, lethargy, convulsions, nausea, vomiting, and dizziness. Seek medical attention immediately if you develop any side effects. But B17-Pro™ is a low-dose product, and it is highly unlikely those symptoms will develop if taken as directed (one tablet per day, or as recommended by your physician, avoiding the foods listed above).
We have just covered a lot of territory about amygdalin, from its history steeped in controversy and politics, to its various mechanisms of action, to an overview of the animal and human studies performed in the 1970’s and early 1980’s that culminated with the one (and only) human efficacy trial, the conclusion of which ended the possibility of further clinical testing. But despite that grim assessment, interest in amygdalin has been reawakened over the past dozen years and a slew of positive studies using cell cultures has demonstrated its potential to inhibit a range of cancer types. Perhaps ongoing research will finally and definitively establish amygdalin’s role in cancer prevention and treatment.
References
- Petersen JC, Markle GE. Politics and science in the Laetrile controversy. Soc Stud Sci. 1979 May;9(2):139-66.
- Dorr RT, Paxinos J. The current status of laetrile. Ann Intern Med. 1978 Sep;89(3):389-97.
- PDQ Integrative, Alternative, and Complementary Therapies Editorial Board. Laetrile/Amygdalin (PDQ®): Health Professional Version. 2017 Mar 15. In: PDQ Cancer Information Summaries. National Cancer Institute (US); 2002-.
- Newmark J, Brady RO, Grimley PM, Gal AE, Waller SG, Thistlethwaite JR. Amygdalin (Laetrile) and prunasin beta-glucosidases: distribution in germ-free rat and in human tumor tissue. Proc Natl Acad Sci U S A. 1981 Oct;78(10):6513-6.
- Ames MM, Moyer TP, Kovach JS, Moertel CG, Rubin J. Pharmacology of amygdalin (laetrile) in cancer patients. Cancer Chemother Pharmacol. 1981;6(1):51-7.
- Rauws AG, Olling M, Timmerman A. The pharmacokinetics of prunasin, a metabolite of amygdalin. J Toxicol Clin Toxicol. 1982 Oct;19(8):851-6.
- Kochi M, Takeuchi S, Mizutani T, Mochizuki K, Matsumoto Y, Saito Y. Antitumor activity of benzaldehyde. Cancer Treat Rep. 1980 Jan;64(1):21-3.
- Milazzo S, Horneber M. Laetrile treatment for cancer. Cochrane Database Syst Rev. 2015 Apr 28;(4):CD005476.
- Lerner IJ. Laetrile: a lesson in cancer quackery. CA Cancer J Clin. 1981 Mar-Apr;31(2):91-5.
- Ellison NM, Byar DP, Newell GR. Special report on Laetrile: the NCI Laetrile Review. Results of the National Cancer Institute’s retrospective Laetrile analysis. N Engl J Med. 1978 Sep 7;299(10):549-52.
- https://www.cancertutor.com/laetrile
- Sabelli R, Iorio E, De Martino A, et al. Rhodanese-thioredoxin system and allyl sulfur compounds. FEBS J. 2008 Aug;275(15):3884-99.
- Bernacki RJ, Niedbala MJ, Korytnyk W. Glycosidases in cancer and invasion. Cancer Metastasis Rev. 1985;4(1):81-101.
- Chen X, Wu B, Wang PG. Glucuronides in anti-cancer therapy. Curr Med Chem Anticancer Agents. 2003 Mar;3(2):139-50.
- Liczbiński P, Bukowska B. Molecular mechanism of amygdalin action in vitro: review of the latest research. Immunopharmacol Immunotoxicol. 2018 Feb 28:1-7.
- Wodinsky I, Swiniarski JK. Antitumor activity of amygdalin MF (NSC-15780) as a single agent and with beta-glucosidase (NSC-128056) on a spectrum of transplantable rodent tumors. Cancer Chemother Rep. 1975 Sep-Oct;59(5):939-50.
- Laster WR Jr, Schabel FM Jr. Experimental studies of the antitumor activity of amygdalin MF (NSC-15780) alone and in combination with beta-glucosidase (NSC-128056). Cancer Chemother Rep. 1975 Sep-Oct;59(5):951-65.
- Ovejera AA, Houchens DP, Barker AD, Venditti JM. Inactivity of DL-amygdalin against human breast and colon tumor xenografts in athymic (nude) mice. Cancer Treat Rep. 1978 Apr;62(4):576-8.
- Stock CC, Tarnowski GS, Schmid FA, Hutchison DJ, Teller MN. Antitumor tests of amygdalin in transplantable animal tumor systems. J Surg Oncol. 1978;10(2):81-8.
- Manner HW, DiSanti SJ, Maggio MI, et al. Amygdalin, vitamin A and enzyme induced regression of murine mammary adenocarcinomas. J Manipulative Physiol Ther. 1978;1(4):246-8.
- https://www.secondopinionfilm.com
- Moertel CG, Ames MM, Kovach JS, Moyer TP, Rubin JR, Tinker JH. A pharmacologic and toxicological study of amygdalin. JAMA. 1981 Feb 13;245(6):591-4.
- Moertel CG, Fleming TR, Rubin J, et al. A clinical trial of amygdalin (Laetrile) in the treatment of human cancer. N Engl J Med. 1982 Jan 28;306(4):201-6.
- Krashen S. Does Laetrile Work? Another Look at the Mayo Clinic Study (Moertel et al., 1982). Internet Journal Alt Med. 2008;7(1).
- Chang HK, Shin MS, Yang HY, et al. Amygdalin induces apoptosis through regulation of Bax and Bcl-2 expressions in human DU145 and LNCaP prostate cancer cells. Biol Pharm Bull. 2006 Aug;29(8):1597-602.
- Park HJ, Yoon SH, Han LS, et al. Amygdalin inhibits genes related to cell cycle in SNU-C4 human colon cancer cells. World J Gastroenterol. 2005 Sep 7;11(33):5156-61.
- Zhou C, Qian L, Ma H, et al. Enhancement of amygdalin activated with β-D-glucosidase on HepG2 cells proliferation and apoptosis. Carbohydr Polym. 2012 Sep 1;90(1):516-23.
- Chen Y, Ma J, Wang F, et al. Amygdalin induces apoptosis in human cervical cancer cell line HeLa cells. Immunopharmacol Immunotoxicol. 2013 Feb;35(1):43-51.
- Makarević J, Tsaur I, Juengel E, et al. Amygdalin delays cell cycle progression and blocks growth of prostate cancer cells in vitro. Life Sci. 2016 Feb 15;147:137-42.
- Juengel E, Afschar M, Makarević J, et al. Amygdalin blocks the in vitro adhesion and invasion of renal cell carcinoma cells by an integrin-dependent mechanism. Int J Mol Med. 2016 Mar;37(3):843-50.
- Juengel E, Thomas A, Rutz J, et al. Amygdalin inhibits the growth of renal cell carcinoma cells in vitro. Int J Mol Med. 2016 Feb;37(2):526-32.
- Makarević J, Rutz J, Juengel E, et al. Amygdalin influences bladder cancer cell adhesion and invasion in vitro. PLoS One. 2014 Oct 15;9(10):e110244.
