GHRP performance (the role of growth hormone releasing peptides)
A lot of people reading this are I am sure, aware of the ‘power’ of growth hormone (GH); a hormone that is secreted by the pituitary gland (see figure 1). GH is popular by those interested in antiaging medicine, especially since Dr. Daniel Rudman’s landmark published article in 1990 in which it was stated; “growth hormone reversed aspects of aging in some individuals by as much as 20-years.” (1)
What Rudman discovered was that by injecting his patients, (aged between 61 and 81) with recumbent human GH, (which remember in the late 1980s had only just recently become commercially available via newly developed production methods), that there were many reversed ‘consequences’ of aging.
Benefits included more elastic and thicker skin, greater hair growth rate and quality, enhanced feelings of wellbeing and of course, perhaps the most famous attributes- decreased body fat levels and more lean muscle mass.
Figure 1: The pituitary gland in relation to the brain.
But as Dr. Richard Walker discussed with me in his interview regarding sermorelin last year (2); today we know that there are some ‘issues’ surrounding GH- when it is used in its recombinant form via subcutaneous injections; these include:
-
- GH needs to be injected to be effective- not everyone wants to inject daily.
-
- GH is somewhat temperature sensitive and needs to be stored in the fridge.
-
- GH creates a negative feedback upon the pituitary gland, which means that if levels are not administered or monitored properly, then it could shut down/ reduce one’s own endogenous production of GH.
-
- GH administered as a bolus injection does not mimic the natural pulsate release of GH, but rather produces a large bell curve in blood after administration.
-
- GH injections remain relatively expensive.
- GH has become a controlled substance in many countries. This means that its possession without a prescription can be considered to be a felony, and laws regarding import and export require import certificates to be supplied before it can be exported. On top of all this, MasterCard and Visa are now refusing to accept credit card payments for controlled substances, (and some other non-controlled medicines too!) Thus, in this last section, I have explained to you why GH has disappeared from most pharmacy sites.
But not all hope is lost, since there are genuine GH precursors known as Growth Hormone Releasing Peptides (GHRPs) that are real-world alternatives.
IGF-1
Just before we move onto the GHRPs, mention should be made of IGF-1 (insulin like growth factor), which has been documented by Dr. Richard Lippman in his articles on the IAS website (3).
IGF-1 is considered to be the ‘active’ agent that instructs cells to start ‘burning’ fat reserves. Indeed, the laboratory measure for determining the blood level of GH actually ‘looks’ for the levels of IGF-1. When IGF-1 levels are high then so have been GH and vice versa.
In Dr. Ronald Klatz’s book; ‘Grow young with HGH’ (4) -published in 1998, I was quoted in it as saying of IGF-1; “that it will be the GH of choice in a few years’ time.” My thinking was that as IGF-1 became commercially available its price would begin to drop dramatically, (as had been the case with GH). Well, we’re now 15-years on from that statement and today there are a few commercially available IGF-1 products on the market, but unfortunately whilst their price has declined significantly, they remain expensive, so alas IGF-1 still remains the domain of the dedicated elite.

Figure 2: Dr. Ronald Klatz’s book, Grow Young with HGH, released in 1998.
So what else has recently become commercially available that looks promising to improve GH secretion?
GHRPs to the rescue!
Ghrelin, a name derived from ‘growth hormone release inducing’ is a substance that stimulates growth hormone stimulatory receptors or GHSR, (5, 9). Ghrelin is produced primarily in the stomach and upper intestines (6) – but note that whilst GHSR’s are concentrated in the pituitary and hypothalamus, they are also widely distributed in central and peripheral tissues, thus helping to explain the diverse action of GH on the body (6, 10).
But the problem with ghrelin is that it makes you hungry! In fact, it has been dubbed ‘the hunger hormone’ (7) – and there are many blogs on the internet that report this ‘side effect.’ This effect has also been published as occurring in both humans and animals (8). Ghrelin levels are high before eating increasing appetite and low after eating (see figure three). Ghrelin stimulates the appetite with the purpose of increasing the intake of food and promoting the storage of fat. So when ghrelin levels are high, we feel hungry. After we eat, ghrelin levels fall and we feel satisfied. Thus ghrelin may not be the supplement of choice for those wishing to lose weight, although it may be the one for those who want the desire to eat more!
However an analogue of ghrelin, known as GHRP2 can realise the GH benefits of ghrelin without triggering any significant hunger pains (11).
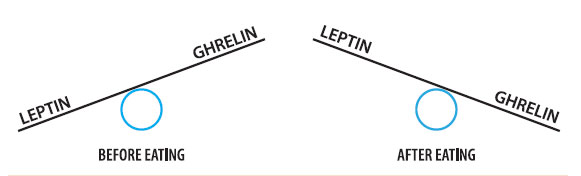
Figure 3: The relationship between ghrelin and leptin before and after eating. Leptin and ghrelin have a ‘teeter-totter’ relationship. When leptin levels rise, ghrelin levels fall. Likewise, when ghrelin levels rise, leptin levels fall.
GHRP2 clinically studied
One placebo controlled study with GHRP2 (12) on 94 healthy individuals (male and female) aged between 40 and 70 years of age took various measures including waist and hips and ratios, total body composition (fat, water, bone and muscle mass), and forced lung capacity at its commencement.
The subjects received either 10 ml of GHRP2 or a placebo each morning for 90 consecutive days. At the end of the study it was shown that those using GHRP2 improved their IGF-1 levels, from an average (pre-study) of 103.54 ng/ml to an average of 120.47 ng/ml (post study), thus clearly indicating that GH levels had been improved; however those using placebo had no significant changes in their IGF-1 (hence GH) levels.
These improved GH levels did, (as expected) in the GHRP2 patient group alter various physical parameters. For example, total body fat decreased on average by 9.14% with visceral fat levels decreasing on average by 14.27% (with no significant changes in the placebo group).
At the same time, muscle mass (as lean body mass) increased on average by 5.37% in the GHRP2 group; plus, forced lung capacity was increased on average by 16.61% (and again there no significant changes in the placebo group).
All these positive changes to these aging biomarkers corroborate findings from the studies that utilized GH injections.
Side effects
Few side effects were noted in the study (12), with just some soft stools and some upset stomachs recorded. There was also an increase in the total blood platelet count, but the values remained within the normal clinical range.
Animal studies regarding the mutagenicity of GHRP2, as well as its LD50 and various genotoxicity tests have been undertaken (13, 14, 15, 16, 17). None of these studies have shown any reason for concern, despite doses having been administered as high as 2000 mg per Kg bodyweight for 2-days and at 50 mg per Kg bodyweight for 90 consecutive days. Compare this with a typical recommended dose for humans at 4 mg (total bodyweight) per day!
It is worth noting that one quite unusual study administered GHRP2 to children for 8 consecutive month; its outcome lacked any significant side-effects or toxicities (18). Another children reference also showed safety after 12 months of GHRP2 administration BID, (J Pediatr Endocrinol Metab. 2003 Sep; 16(7):981-5).
Uses
GHRP2 is clearly a useful adjunct to boost GH levels. Therefore, perhaps its most direct application is to assist in the age-related disorder of sarcopenia.
Sarcopenia is the loss of lean body mass. Surprisingly, muscle loss can begin early in life, even by 25-years of age. Clinical studies that measure the excretion of creatinine, (a measure of muscle content) note decreases of 50% between the ages of 25 and 70. This fact highlights that the degradation of muscle exceeds its synthesis past young adulthood.
Clearly, maintaining muscle mass is essential to supporting whole-body protein metabolism, wound healing, physical strength, organ function, skin integrity and immunity. In the elderly, the loss of lean body mass manifests itself as frailty, reduced ability to walk, climb stairs, rise from a chair etc. This loss of independence can not only lead to disability but depression too.
As a side note, some fairly recent studies suggest that patients with growth hormone deficiency have a higher risk of developing dental diseases (19). Another dental study utilised GH around implants (20), it recorded that bone repair and collagen fibers were denser even 12 weeks after surgery in those who used GH. It was stated that progressive insufficiency of GH during aging may contribute to periodontal disease and tooth loss.
Therefore, GHRP2 supplementation could benefit the maladaptive effects of sarcopenia on an individual’s body composition and extend good dental health.
GHRP2 synergy
Sermorelin is another very fascinating GH peptide and was described by Dr. Richard Walker in issue 2 of the 2012 Aging Matters™ magazine. Sermorelin acts as a secretagogue to encourage the release of GH from the pituitary gland into the blood and it has been shown to be efficacious when taken sublingually, and it is stable at room temperature.
Dr. Walker told me that when GHRP2 is taken in combination with sermorelin that the total effects of GH release can be enhanced by up to five-fold! Studies have confirmed this combination synergy, (21, 22, 23, 24, 25, 26). This is obviously a massive improvement and suggests that the combination of these two peptides, (sermorelin taken sublingually PM and GHRP2 taken orally AM) could provide a genuine alternative to injectable GH.
GHRP2-Pro™
IAS’s has now introduced GHRP2 into its peptide range under the name of GHRP2-Pro™. It is an oral liquid, stable at room temperature that can be taken at 4 ml (3/4 of a teaspoon) first thing in the morning; (each serving contains 4 mg of GHRP2).
In addition to GHRP2, the GHRP2-Pro™ contains the following supportive agents:
Leucine, (as HMB) is an insulin secretagogue with the capacity to stimulate muscle protein synthesis.
Vitamin D: An essential agent for muscle growth and development.
Mucuna pruriens: A legume extract that mimics L-dopa to increase dopamine levels to enhance GH production.
Conclusion
Proper GHRP therapies are perhaps not yet ‘cheap’ options, but at least they are now affordable by many more people. We’ve experienced years of all kinds of touted GH ‘alternatives’ many of which were at best weak performers, or at worst virtual complete wastes of time and money. But now at last, the marketplace has genuine GH alternatives that do not have to be injected to be effective.
Thus, there’s mounting evidence that a combination of lifestyle, exercise and anabolic ‘medical food’ supplements such as GHRP2-Pro™ can prevent and reverse sarcopenia in older persons, therefore guarding against this cause of morbidity and mortality.
References
1. Rudman, D. et al, ‘Effects of growth hormone in men over 60 years old.’ New England Journal of Medicine, Volume 323, 1-6, July 5, 1990
2. Walker, R., ‘Sermorelin, is it better than growth hormone?’ Aging Matters™ magazine, No 2, 2012
3. Lippman, R., www.antiaging-systems.com/articles (choose list by author), June 2013
4. Klatz, R., Khan, C., ‘Grow young with HGH’ Harper Books, May 8, 1998
5. Howard AD et al, ‘A receptor in pituitary and hypothalamus that functions in growth hormone release.’ Science 273: 974-977, 1996
6. Van der Lely AJ et al, ‘Biological, physiological, pathophysiological and pharmacological aspects of ghrelin.’ Endocrine reviews 25(3); 426-457, 2004
7. Carol Kopp, ‘The hunger hormone’ CBS News, February 11, 2009 www.cbsnews.com
8. Wren AM et al, ‘Ghrelin enhances appetite and increases food intake in humans.’ J Clin Endocrinol Metab. 2001 Dec; 86(12):5992.
9. Arvat E et al, ‘Preliminary evidence that ghrelin, the natural GH secretagogue (GHS0- receptor ligand, strongly stimulates GH secretion in humans.’ J Endocrinol Invest 23: 493-495, 2000.
10. Papotti M et al, ‘Growth hormone secretagogue binding sites in peripheral tissues.’ J Clin Endocrinol Metab 85: 3803-3807, 2000
11. Mericq V., et al, ‘Changes in appetite and body weight in response to long-term oral administration of ghrelin agonist GHRP2 in growth hormone deficient children.’ J Pediatr Endocrinol Meta, 2003, Sep; 16(7); 981-5.
12. A phase III, randomized, open label, placebo controlled clinical study to evaluate the efficacy and safety of GHRP2 for the treatment of age related changes in form, function and quality of life in healthy subjects. Date on file, ProSoma FLA, 2 March, 2012.
13. Mutagenicity study of GHRP2 by bacterial reverse mutation assay, data on file ProSoma FLA, April 18, 2012.
14. Mammalian bone marrow chromosome aberration test of GHRP2 in Swiss albino mice. Data on file, ProSoma FLA, 1 May, 2012.
15. Acute oral toxicity study of GHRP2 in Wistar rats. Data on file, ProSoma FLA, 19 August, 2011.
16. 90 day oral toxicity study of GHRP2 in Wistar rats. Data on file, ProSoma FLA, 16 June, 2011.
17. Micronucleus test of GHRP2 in Swiss albino mice. Data on file, ProSoma FLA, 18 April, 2012.
18. Mericq V et al, ‘Effects of eight months treatment with graded doses of growth hormone releasing peptide in GH deficient children.’ J Clin Endocrinol Meta, 83: 2355-2360, 1998.
19. Britto IM et al, ‘Periodontal disease in adults with untreated congenial growth hormone deficienct; a case control study.’ J Clin Periondontal, 2011 Jun; 38(6): 525-31.
20. Hossan E et al, ‘Histological evaluation of the effect of using growth hormone around immedediate dental implants in fresh extraction sockets.’ Implant dentistry, 20 (1): 47-55, 2011.
21. Argente J. et al, ‘Growth hormone releasing peptides; clinical and basic aspects.’ Horm Res 1996; 46 (4-5): 155-9
22. Ghigo E., et al, ‘Growth hormone releasing peptides.’ Eur J Endocrinol, 1997 May, 136(5): 445-60.
23. Ghigo E., et al. ‘Biologic activities of growth hormone secretagogues in humans.’ Endocrine, 2001 Feb; 14(1): 87-93.
24. Root AW., et al, ‘Clinical pharmacology of human growth hormone and its secretagogues.’ Curr Drug targets immune Endocr Metabol Disord, 2002 Apr; 2(1): 27-52.
25. Cunha SR. et al, ‘Ghrelin and growth hormone secretagogues potentiate GH releasing hormone induced cyclic adenosine 3,5 monophosphate production in cells expressing transfected GHRH and GH secretagogue receptors.’ Endocrinology, 2002 Dec, 143(12):4570-82.
26. Casanueva FF. et al, ‘Growth hormone releasing hormone as an agonist of the ghrelin receptor GHS-R1a.’ Proc Natl Acad Sci USA, 2008 Dec 23; 105(51): 20452-7.
