Balancing Hormones in Men over 40 with Anastro Pro™ and DIM Pro2
As most longevity enthusiasts are aware, aging leads to significant alterations in many of the body’s systems, including endocrine balance. In both women and men, advancing years are associated with declining sex hormone production; in women, the fall in estrogen occurs at a rapid, often dramatic rate around menopause, while in men, testosterone levels wane slowly and steadily over the years. Much attention is directed to supplementing female hormones for symptom relief during menopause, but the male equivalent of hormonal aging, known as “andropause,” often also requires treatment and modulation ─ the focus of this article. We’ll discuss how to restore male hormones to youthful levels by preventing an age-related enzymatic reaction that depletes testosterone by converting it to estrogen, and discover that inhibiting this enzyme with the drug anastrozole (Anastro Pro™) boosts one’s endogenous testosterone production ─ a novel approach to testosterone replacement therapy ─ while suppressing excess estrogen. Once estrogen is in the normal range, we’ll also learn how to optimize its metabolism with Dim Pro2™, which offers a wealth of cancer-preventing and health-promoting benefits.
The Dangers of Low Testosterone Coupled with High Estrogen
Testosterone is important in maintaining a variety of essential metabolic functions, and age-related deficits are associated with increased risk of atherosclerosis, (1,2) cardiovascular disease, (3,4) and prostate cancer; (5-7) decreased lean body mass; (8,9) increased abdominal fat; (8,10-12) osteoporosis; (8,13) decreased libido; (8) diabetes and insulin resistance; (10-12,14) depression (15) and cognitive deficits. (8,16) Although several factors may contribute to this hormonal shortfall, a major facilitator is the increased activity of the enzyme aromatase, (8) which converts testosterone to a major form of estrogen (estradiol):
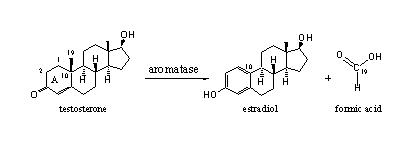
(Source: Ergogenics.org)
Overweight men often exhibit this imbalanced ratio of high estrogen to low testosterone as evidenced by the development of breast tissue. A metabolic vicious circle is initiated by low testosterone, which results in increased abdominal fat, and since belly fat serves as an ideal factory for the manufacture of large quantities of aromatase, (17) testosterone becomes further suppressed, boosting estrogen even higher, fueling the hormonal imbalance. Although it’s beneficial to maintain estrogen levels within the normal range for males, elevated estrogen, like low testosterone, can increase the risk of heart attack, (18-20) prostate cancer, (21) and other conditions. We’ll see below the potentially dangerous effects if some of this estrogen metabolizes to a particularly potent form, 16-α-hydroxyestrone.
Anastrozole: A Novel Approach to Testosterone Replacement
Ideally, the goal is to maintain levels of testosterone and estrogen equivalent to those of a young, healthy man. An effective and clinically proven method to accomplish this is by inhibiting the action of aromatase with the drug anastrozole (Anastro Pro™). Anastrozole is used to treat breast cancer in women, (8,13,22) and as a novel means of normalizing testosterone and estrogen levels in aging men. (8,13)
Levels of testosterone, like other hormones, are constantly monitored by the hypothalamus in the brain; normally, when levels are low, the hypothalamus signals the pituitary to step up gonadal production. But in aging men, excess estrogen saturates testosterone receptor cites in the brain, altering the normal feedback mechanism and shutting down testosterone production. By suppressing estrogen, anastrozole reduces that hormone’s negative feedback on the brain and instead stimulates the pituitary to increase endogenous testosterone production to the mid-normal range of a young healthy man. (8,13) The result is the desired increase in the circulating testosterone to estrogen ratio.
Of course, one could simply supplement with prescription testosterone. But the many forms of testosterone replacement, such as oral tablets and injections, have limitations according to some researchers, including its safety in older men, (8,13) not to mention the fact that supplemental testosterone could potentially be converted to estrogen if aromatase levels are high, further exacerbating the hormonal imbalance. Anastrozole provides an alternative to testosterone injections, pills or patches, and according to researchers, is “a well-tolerated orally administered agent [that] may have unique potential as a means of adrogen replacement therapy.” (8)
Anastrozole Clinically Proven to Restore Youthful Hormone Balance
Several studies have been conducted to evaluate the drug’s effect in suppressing aromatase and improving male hormone levels, including a placebo-controlled double-blind study in men aged 62-74 with low or borderline-low testosterone levels. Subjects received either 1 mg anastrozole daily (the dose used in female breast cancer patients), 1 mg anastrozole twice a week, or placebo. In each of the treated groups, testosterone levels were restored to a youthful normal range, and the gains were only slightly higher in the 1-mg/day group than in the lower-dose group. For example, mean bioavailable testosterone (i.e., testosterone that is either free or loosely bound to blood protein) increased from 99 to 207 ng/dl in the 1-mg/day group and 115 to 178 in the lower-dose group. Estradiol was lowered to a favorable level, 17 pg/ml, in each of the treated groups. (8) These results are consistent with other studies that show normalization of hormone balance with anastrozole administration. (13, 23-25)
Anastro Pro™, 100 mcg/tablet, is a low-dose anastrozole therapy. The higher 1-mg dose is the strength often used to treat breast cancer and may cause some adverse effects in men. See “Summary and Recommendations” below.
Aging Men Must Also Balance Estrogen Metabolism
We now know that anastrozole inhibits the aromatization reaction enabling men to reestablish a more youthful testosterone to estrogen profile. But there’s more hormonal fine-tuning to do, because even when maintaining estrogen in the normal range, it is crucial balance its metabolism. (This is true for both men and women.) Let’s see why.
The estrogens are a group of hormones consisting of estradiol, estrone, estriol, and a variety of metabolites. As mentioned, estradiol is the predominant form in circulation, and can metabolize through two different pathways, producing either 2-hydroxyestrone, a beneficial form with anti-proliferative (anti-cancer) effects or 16-α-hydroxyestrone, a particularly potent analog with carcinogenic effects. (For ease of our discussion, we’ll refer to the 2-hydroxy form as “good” estrone and the 16-α-hydroxy form as “bad” estrone.) Of note here is the ratio of these two metabolites, which can be used as a predictive biomarker to asses the risk of developing hormone-responsive cancers such as breast, (26,27) thyroid, (26) and prostate, (28-31) with an increased “good” to “bad” estrone ratio favoring a decreased risk of developing cancer. For example, studies examining urinary estrogen metabolites in relation to prostate cancer suggest that men with the highest “good” to “bad” estrone ratios have a reduced risk of developing cancer. (30-31)
Cruciferous Vegetable Derivative “DIM” Enhances “Good” Estrone and Fights Cancer
So how can we shift the estradiol metabolic pathway in favor of “good“estrone? With the phytochemical 3,3′-diindolylmethane (DIM), a derivative of cruciferous vegetables such as broccoli, cauliflower, and Brussels sprouts. It has long been recognized that these crucifers are full of beneficial compounds, such as indoles, which possess chemopreventive and chemotherapeutic properties against a wide range of cancers. (26, 28-29) Indole-3-carbinol is one such compound; however, it is unstable and dimerizes (i.e., forms a molecular double image of itself) into DIM (26, 28-29) under certain conditions, such as during digestion and cooking, establishing DIM as the dominant active (and stable) indole from cruciferous vegetables. (28)
A study evaluating the efficacy of DIM in thyroid proliferative disease found that 300 mg DIM increased the ratio of “good“ to “bad” estrone by 170 % in two weeks. (26) These results are consistent with another study in which 150 mg DIM increased the “good” to “bad” estrone ratio by 76% in patients with benign and pre-cancerous Human papillomavirus. (32) DIM has been demonstrated to protect against several types of cancers such as breast, (27,29) prostate, (29, 33-34) and thyroid, (26) by counteracting the adverse effects of “bad“ estrone. DIM induces apoptosis (“programmed cell death”) (26,28) and inhibits angiogenesis (the growth of new blood vessels), (35) leading to decreased cancer cell growth and proliferation.
DIM is a unique therapy that optimizes estrogen metabolism and reduces the risk of cancer, without side effects. DIM is available as DIM- Pro2™, an updated version with greater potency (100 mg) than the previous product.
Summary and Recommendations
The combination of Anastro Pro™ and DIM Pro™ is a unique dual-prong approach to normalizing age-related alterations in hormone balance by (1) restoring youthful testosterone levels while suppressing estrogen overproduction and (2) preventing cancer by favorably influencing estrogen metabolism. Although DIM Pro™ use can be initiated at any time, medical supervision and blood testing, including PSA, free and total testosterone, estradiol, and DHEA, are recommended before and during Anastro-Pro™ therapy. And of course, a healthy diet coupled with a suitable exercise program that includes ample strength training, will support your overall goal of optimizing and restoring hormone balance.
References
1. Hak AE, Witteman JC, de Jong FH, et al. Low levels of endogenous androgens increase the risk of atherosclerosis in elderly men: the Rotterdam study. J Clin Endocrinol Metab. 2002 Aug;87(8):3632-9.
2. Jones RD, Nettleship JE, Kapoor D, Jones HT, Channer KS. Testosterone and atherosclerosis in aging men: purported association and clinical implications. Am J Cardiovasc Drugs. 2005 5(3):141-54.
3. Malkin CJ, Pugh PJ, Morris PD, Asif S, Jones TH, Channer KS. Low serum testosterone and increased mortality in men with coronary heart disease. Heart. 2010 Nov;96(22):1821-5.
4. Ohlsson C, Barrett-Connor E, Bhasin S, et al. High serum testosterone is associated with reduced risk of cardiovascular events in elderly men. J Am Coll Cardiol. 2011 Oct 11;58(16):1674-81.
5. Morgentaler A, Rhoden EL. Prevalence of prostate cancer among hypogonadal men with prostate-specific antigen levels of 4.0 ng/mL or less. Urology. 2006 Dec;68(6):1263-7.
6. Hoffman MA, DeWolf WC, Morgentaler A. Is low serum free testosterone a marker for high grade prostate cancer? J Urol. 2000 Mar;163(3):824-7.
7. Morgentaler A, Bruning CO 3rd, DeWolf WC. Occult prostate cancer in men with low serum testosterone levels. JAMA. 1996 Dec 18;276(23):1904-6.
8. Leder BZ,Rohrer JL, Rubin SD, Gallo J, Longcope C. Effects of aromatase inhibition in elderly men with low or borderline-low serum testosterone levels. J Clin Endocrinol Metab. 2004 Mar;89(3):1174-80.
9. Yuki A, Otsuka R, Kozakai R, et al. Relationship between Low Free Testosterone Levels and Loss of Muscle Mass. Sci Rep. 2013 May 10;3:1818.
10. Chen RY, Wittert GA, Andrews GR. Relative androgen deficiency in relation to obesity and metabolic status in older men. Diabetes Obes Metab. 2006 Jul;8(4):429-35.
11. Abate N, Haffner SM, Garg A, Peshock RM, Grundy SM. Sex steroid hormones, upper body obesity, and insulin resistance. J Clin Endocrinol Metab. 2002 Oct;87(10):4522-7.
12. Boyanov MA, Boneva Z, Christov VG. Testosterone supplementation in men with type 2 diabetes, visceral obesity and partial androgen deficiency. Aging Male. 2003 Mar;6(1):1-7.
13. Burnett-Bowie SA, McKay EA, Lee H, Leder BZ. Effects of aromatase inhibition on bone mineral density and bone turnover in older men with low testosterone levels. J Clin Endocrinol Metab. 2009 Dec;94(12):4785-92.
14. Grossmann M, Thomas MC, Panagiotopoulos S, et al. Low testosterone levels are common and associated with insulin resistance in men with diabetes. J Clin Endocrinol Metab. 2008 May;93(5):1834-40.
15. Almeida OP, Yeap BB, Hankey GJ, Jamrozik K, Flicker L. Low free testosterone concentration as a potentially treatable cause of depressive symptoms in older men. Arch Gen Psychiatry. 2008 Mar;65(3):283-9.
16. Hogervorst E, Bandelow S, Combrinck M, Smith AD. Low free testosterone is an independent risk factor for Alzheimer’s disease. Exp Gerontol. 2004 Nov-Dec;39(11-12):1633-9.
17. Kalyani RR, Dobs AS. Androgen deficiency, diabetes, and the metabolic syndrome in men. Cur Opin in Endocrinol Diab Obesity. 2007; 14: 226-234.
18. Mohamad MJ, Mohammad MA, Karayyem M, Hairi A, Hader AA. Serum levels of sex hormones in men with acute myocardial infarction. Neuro Endocrinol Lett. 2007 Apr;28(2):182-6.
19. Cao J, Zou H, Zhu BP, et al. Sex hormones and androgen receptor: risk factors of coronary heart disease in elderly men. Chin Med Sci J. 2010 Mar;25(1):44-9.
20. Barud W, Palusinski R, Beltowski J, et al. Relation between markers of inflammation and estradiol in older men. Med Sci Monit. 2010 Dec;16(12):CR593-7.
21. Singh PB, Matanhelia SS, Martin FL. A potential paradox in prostate adenocarcinoma progression: oestrogen as the initiating driver. Eur J Cancer. 2008 May;44(7):928-36.
22. Plourde PV, Dyroff M, Dukes M. Arimidex: a potent and selective fourth-generation aromatase inhibitor. Breast Cancer Res Treat. 1994;30:103–111.
23. Mauras N, O’Brien KO, Klein KO, Hayes V. Estrogen suppression in males: metabolic effects. J Clin Endocrinol Metab. 2000;85:2370–2377.
24. Raman JD, Schlegel PN. Aromatase inhibitors for male infertility. J Urol. 2002;167:624–629.
25. Taxel P, Kennedy DG, Fall PM, Willard AK, Clive JM, Raisz LG. The effect of aromatase inhibition on sex steroids, gonadotropins, and markers of bone turnover in older men. J Clin Endocrinol Metab. 2001; 86:2869–2874.
26. Rajoria S, Suriano R, Parmar PS, et al. 3,3′-diindolylmethane modulates estrogen metabolism in patients with thyroid proliferative disease: a pilot study. Thyroid. 2011 Mar;21(3):299-304.
27. Meilahn EN, De Stavola B, Allen DS, et al. Do urinary estrogen metabolites predict breast cancer? Follow up of the Guernsey III cohort. Br J Cancer. 1998;78:1250–1255.
28. Heath EI, Heilbrun LK, Li J, et al. A phase I dose-escalation study of oral BR-DIM (BioResponse 3,3′- Diindolylmethane) in castrate-resistant, non-metastatic prostate cancer.Am J Transl Res. 2010 Jul 23;2(4):402-11.
29. Le HT, Schaldach CM, Firestone GL, Bjeldanes LF. Plant-derived 3,3′-Diindolylmethane is a strong androgen antagonist in human prostate cancer cells. J Biol Chem. 2003 Jun 6;278(23):21136-45.
30. J Barba M, Yang L, Schünemann HJ, et al. Urinary estrogen metabolites and prostate cancer: a case-control study and meta-analysis. Exp Clin Cancer Res. 2009 Oct 8;28:135.
31. Muti P, Westerlind K, Wu T, et al. Urinary estrogen metabolites and prostate cancer: a case-control study in the United States. Cancer Causes Control. 2002 Dec;13(10):947-55.
32. Zeligs MA, Sepkovic DW, Manrique C, Macsalka M, Williams DE, Bradlow HL. Absorption-enhanced 3,3-diindolylmethane: human use in HPV-related, benign and pre-cancerous conditions. 2002;Proc Am Assoc Cancer Res. 43:3198.
33. Cho HJ, Park SY, Kim EJ, Kim JK, Park JH. 3,3’-Diindolylmethane inhibits prostate cancer development in the transgenic adenocarcinoma mouse prostate model. Mol Carcinog. 2011 Feb;50(2):100-12.
34. Chen D, Banerjee S, Cui QC, Kong D, Sarkar FH, Dou QP. Activation of AMP-activated protein kinase by 3,3’-diindolylmethane (DIM) is associated with human prostate cancer cell death in vitro and in vivo. PLoS One. 2012;7(10):e47186.
35. Chang X, Tou JC, Hong C, et al. 3,3’-Diindolylmethane inhibits angiogenesis and the growth of transplantable human breast carcinoma in athymic mice. Carcinogenesis. 2005 Apr;26(4):771-8.
